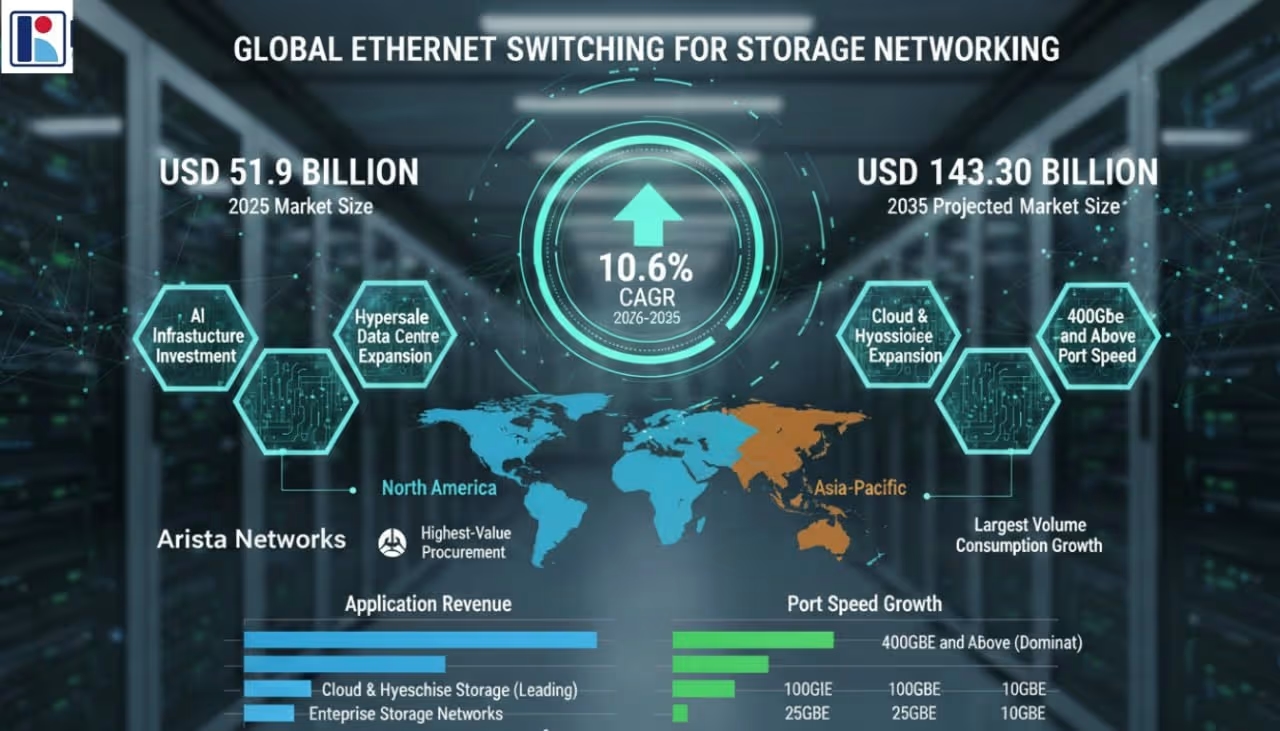
2026-05-22T18:30:00.000Z
Aug 06, 2025 Blog
Every three minutes, someone in the United States is diagnosed with leukemia, lymphoma, or myeloma. Behind that statistic is a treatment landscape that, until a decade ago, offered patients with relapsed or refractory blood cancers very limited options beyond conventional chemotherapy. CAR T-cell therapy is changing that equation—and the scale of unmet need is driving market growth that compounds at rates unusual even by oncology standards.
CAR T-cell therapy works by reprogramming a patient's own immune cells to identify and eliminate cancer. T cells are extracted from the patient, genetically modified to carry chimeric antigen receptors that recognise specific cancer cell markers, and reinfused. In B-cell leukemias, the target is typically CD19—a protein expressed on malignant cell surfaces that the engineered T cells are trained to destroy.
The precision of this approach is what separates it from earlier immunotherapy models. Rather than stimulating the immune system broadly, CAR T-cell therapy directs it toward a defined cellular target, reducing collateral damage and improving outcomes in patient populations where conventional treatment has failed. The clinical evidence supporting its use across multiple blood cancer subtypes—acute lymphoblastic leukemia, diffuse large B-cell lymphoma, multiple myeloma—has grown substantially since the first approved therapies reached the market.
The Global CAR T-Cell Therapy Market was valued at approximately USD 2.04 billion in 2022 and is projected to expand at a CAGR of 39.4% through 2030, according to Bizwit Research & Consulting. That growth rate is a function of two compounding forces: rising global blood cancer incidence and an accelerating pipeline of next-generation therapies.
The Leukemia & Lymphoma Society estimates that new leukemia, lymphoma, and myeloma cases will account for 9.4% of all new cancer diagnoses in the United States in 2023. These are commercially reachable patient populations concentrated in specialised treatment centres—a distribution pattern that aligns with the infrastructure requirements of a high-complexity, high-cost therapy. The market isn't growing despite the treatment's complexity. It's growing in part because of the institutional investment that complexity is attracting.
The capacity to deliver CAR T-cell therapy is expanding alongside the clinical evidence. Fortis Healthcare's January 2024 launch of the Fortis Institute of Blood Disorders integrates paediatric and geriatric treatment, advanced transplant procedures, and hematopathology expertise across multiple locations in Delhi, Mumbai, Bengaluru, Noida, and beyond. The institute offers NexCAR19—India's first commercially approved CAR T-cell therapy.
NexCAR19 was developed by ImmunoACT, an IIT Bombay spin-off, and targets B-cell lymphomas and B-acute lymphoblastic leukemia in patients aged 15 and above. It's indigenously developed, not imported—a distinction that matters in a market where treatment cost has historically been the primary barrier to access. India's first approved CAR T-cell therapy reaching the market through a domestic academic-commercial partnership is a signal about the direction of the global infrastructure buildout.
AbbVie and Umoja Biopharma's January 2024 agreements provide a useful marker of where pipeline capital is concentrating. Under two exclusive option and license agreements, AbbVie secured rights to Umoja's CD19-directed in-situ CAR T-cell programme—including UB-VV111, currently in IND-enabling phase—alongside collaboration rights to develop up to four additional therapy candidates on Umoja's VivoVec platform.
In-situ CAR T-cell production generates the therapeutic cells within the patient's body rather than through an external manufacturing process. The commercial significance is substantial: it has the potential to reduce the cost and logistical complexity of a therapy whose current price point—often exceeding $400,000 per treatment course—remains the primary constraint on wider adoption.
The primary constraints on market growth are well defined: high treatment cost, manufacturing complexity, and the risk of adverse events including cytokine release syndrome. These are real barriers, not footnotes. They are also the targets of the most active areas of R&D investment in the field.
A therapy that enters the 2030s with improved in-situ manufacturing, a broader approved indication set, and a cost structure accessible to more healthcare systems will look substantially different from the market that exists today. The growth trajectory between now and then reflects the distance still to close.
Latest Blogs

2026-05-22T18:30:00.000Z

2026-05-01T18:30:00.000Z

2026-04-17T18:30:00.000Z